
Find out what solar panels cost in your area
A lead acid battery is a kind of rechargeable battery that stores electrical energy by using chemical reactions between lead, water, and sulfuric acid. The technology behind these batteries is over 160 years old, but the reason they’re still so popular is because they’re robust, reliable, and cheap to make and use.
Should you choose lead acid batteries for your home energy storage needs? Probably not.
Lead acid batteries can be somewhat more affordable than newer lithium-based technology, but they are almost certainly more difficult to use and maintain and require more hands-on work and knowledge to get working.
If you’re looking to store energy produced by a solar array, lithium iron phosphate batteries will prove more convenient, compact, and usable. For specific recommendations, check out our guide to the best home solar batteries.
If you’re still interested in how lead acid batteries work for home energy storage, read on!
Key takeaways
-
Lead acid batteries for solar energy storage are called “deep cycle batteries.”
-
Different types of lead acid batteries include flooded lead acid, which require regular maintenance, and sealed lead acid, which don’t require maintenance but cost more.
-
Lead acid batteries are proven energy storage technology, but they’re relatively big and heavy for how much energy they can store.
-
Deep cycle lithium ion batteries are more expensive than nearly all lead acid batteries, but are much more compact and maintenance-free.
How a lead acid battery works
While the chemistry of lead acid batteries is quite simple, writing out all the chemical equations can make it seem very complicated, so we’ll try to explain it without all of that.
The simplest version of a lead acid battery consists of three things:
A metal plate made of lead and antimony with a negative charge
A positively-charged metal plate made of lead dioxide
A mixture of sulfuric acid and water
The negative plate is called an anode, the positive plate is called a cathode, and the water/acid mixture is called an electrolyte.
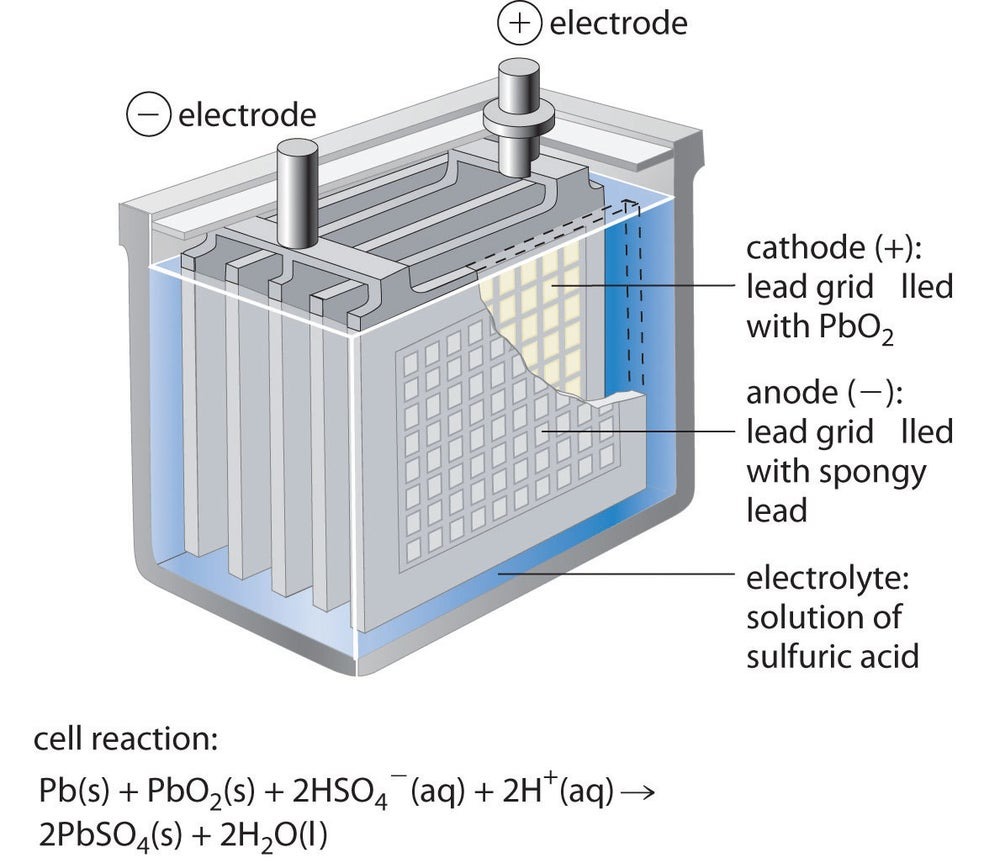
A simple lead acid battery. Image source: LibreTexts.org
When the plates are suspended in the electrolyte mixture and connected to wires, the battery is ready to provide electricity!
As electrons flow out of the battery, the acid in the electrolyte begins to stick to the lead on the electrodes, converting their outer surfaces to lead sulfate and leaving extra hydrogen ions floating in the water.
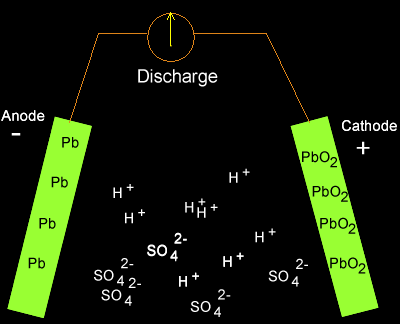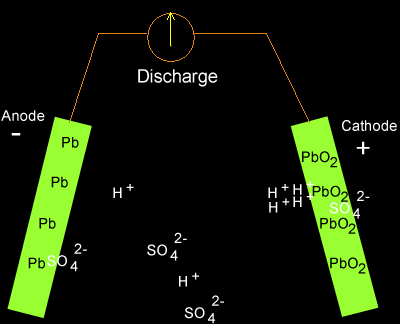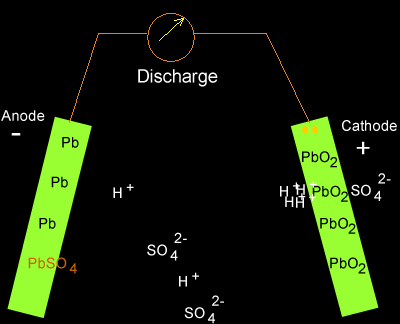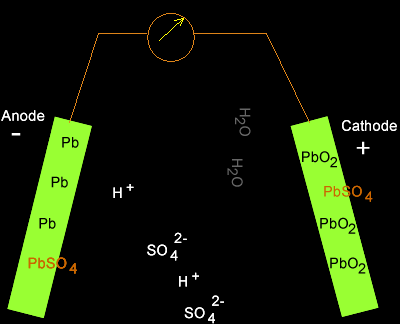
As electrons are sent along the wires, lead sulfate forms on the plates while the battery discharges. Source: Dynamic Science
Charging the battery adds electrons back in and breaks the electrochemical bonds between the lead and sulfate. The sulfate recombines with the free hydrogen ions in the electrolyte to make sulfuric acid again.
There is a drawback to the lead acid design. If the battery is discharged too much, some of the lead sulfate can’t be broken down and recombined with the free hydrogen, which results in a permanent coating on the lead plates called sulfation. Sulfation greatly reduces the lifespan of the battery.
In order for lead acid batteries to work for long periods of time, they must be discharged no more than half of their total battery capacity on a regular basis.
Automotive batteries vs deep cycle batteries
Automotive batteries are not well-suited for storing energy for home use because they are designed to give short bursts of electricity that are used to start a car. In fact, these types of batteries are called starting, lighting, and ignition (SLI) batteries.
SLI batteries are made with thin, porous lead plates that are designed to draw as much electricity as possible from the electrolyte at one time. That surge of energy is called current, and it is measured in amps.
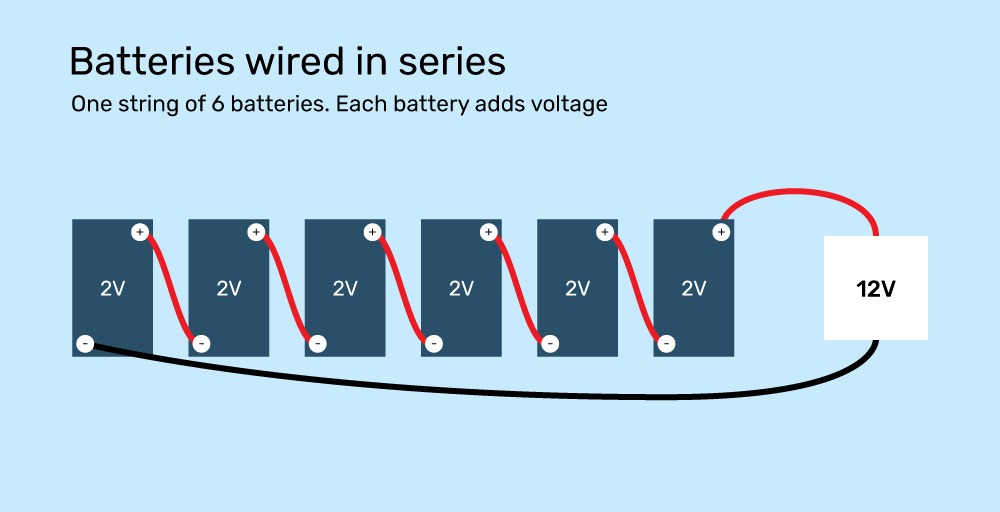
Automotive batteries are made using 6 cells wired in series. When the battery is fully charged, each cell produces around 2 volts, leading to a total voltage of 12 volts. Volts times amps equals wattage, or total power available at one time.
Learn more: Amps, volts, and watts
Deep cycle batteries for solar energy storage don’t have to produce a bunch of instantaneous power to start anything, so they have thicker lead plates that will last a long time and draw power from the electrolyte more slowly and evenly.
Deep cycle battery banks for solar storage
Deep cycle batteries tend to be large rectangular boxes made of a plastic composite material, which makes them easy to stack next to one another. Because they don’t have to start a car, they can produce less wattage individually, and can be wired together to make battery banks.
Many deep cycle batteries for energy storage have only one large cell and produce 2 volts. And, the larger the cell - the more energy it can store. Other 2, 3, and 6-cell designs are found in batteries of 4, 6, and 12 watts, respectively. Battery banks made for storing solar energy are wired together to produce 12, 24, or 48 volts.
For example, six 2-volt batteries can be wired in series (negative to positive all down the line) to make a 12-volt battery bank, or four 12-volt batteries can be wired in series to make a 48-volt battery bank.
If you think of electricity like water, the battery voltage corresponds to the pressure in a hose. Higher pressure means a greater rate of flow. Current, measured in amperage, defines how wide the opening of the hose is. The two together determine wattage, or the rate of flow.
Energy storage capability is measured in amp-hours per 20 hours, which means it’s measured by the amount of energy it would take to deplete the battery in 20 hours. Imagine a hose attached to a barrel. A bigger battery is like a bigger barrel, because it holds more energy (water).
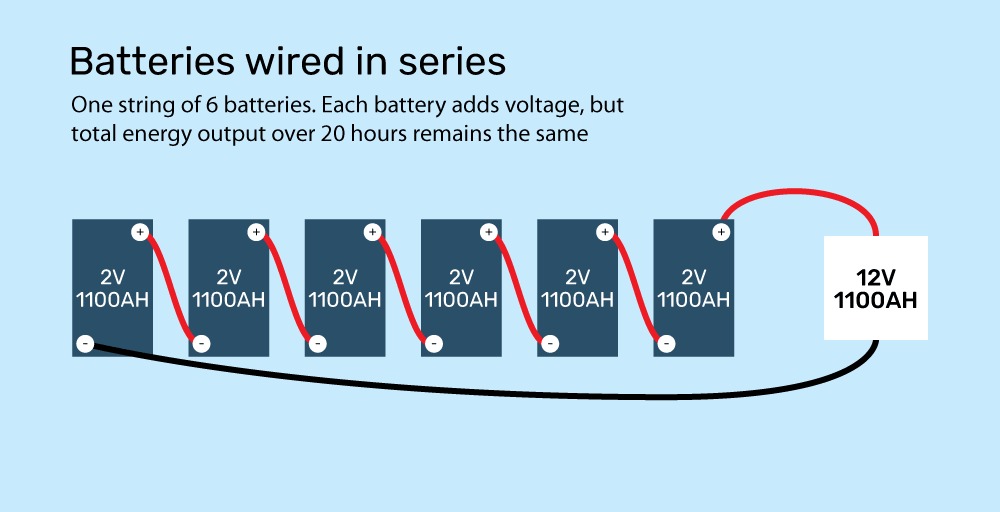
You might see a 2-volt battery that is rated to store 1100 amp-hours. That means the battery can put out 55 amps for 20 hours. At 2 volts, that means the battery would be making 110 watts at any given time (2 volts x 55 amps = 110 watts).
But 2 volts is not very high pressure. It would be like holding the hose level with the barrel. There’s a lot of water behind it, but it just trickles out. The idea is to create more pressure so the energy can flow at a higher rate.
Wiring batteries in series to increase the voltage is like adding pressure to the water. Take the battery bank from earlier: wiring the six 2-volt batteries in series doesn’t increase the energy stored, it just increases how fast it flows by the pressure in the “hose”.
In this case, the series of batteries can still produce 55 amps for 20 hours, but this time they produce it at 12 volts, for a total of 660 watts of power (55 amps times 12 volts).
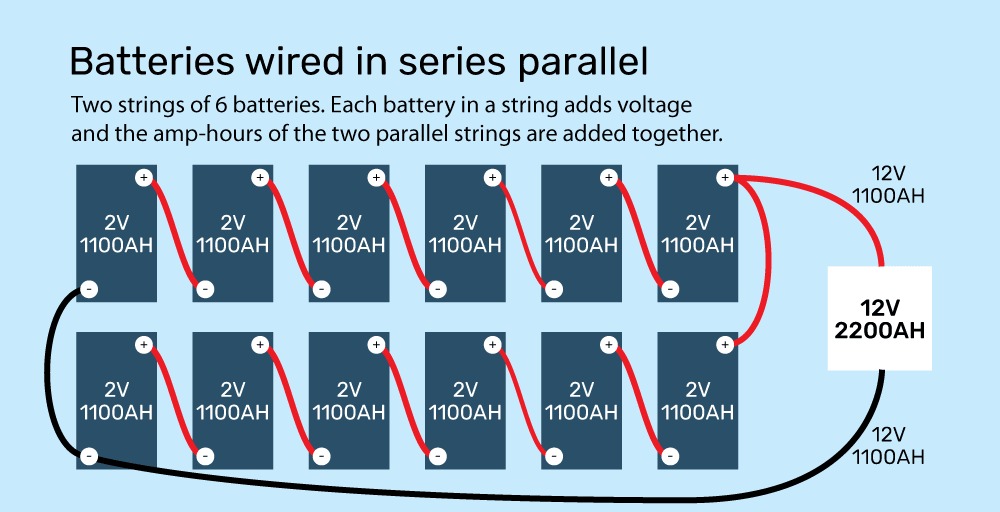
Conversely, wiring two batteries in parallel increases the amperage, but not the voltage. Parallel wiring connects the negative terminals of both batteries and the positive terminals of both batteries. Parallel wiring is acceptable if you’re connecting batteries of 12 volts together, but most people will need a battery bank that can produce 24 or 48 volts; in this case, they will need to wire in series-parallel.
Series-parallel wiring connects two or more strings of batteries wired in series. This increases both the total energy stored as well as the voltage, as seen in the image below.
Designing a battery bank
Designing a battery bank for solar storage is a balancing act of finding the right voltage, the right current, and the right amount of stored energy.
Most homes need a total of around 900 kilowatt hours (kWh) of electricity per month, or 30 kWh per day. To serve those needs, you’d need a battery bank capable of storing 625 amp-hours at 48 volts.
An important rule of designing a battery bank is to not allow a current of more than 100 amps. The main idea here is to build a battery bank with a high enough voltage so that the current will not exceed 100 amps.
Battery voltage drops as the battery is reduced from a full charge, so it’s important to take that into consideration when planning your battery bank. If you plan to run a lot of appliances on battery power alone, it’s a good idea to choose a 48-volt battery bank so you can draw as much as 4,800 watts at a time.
When you are using home appliances, each one requires a certain amount of power, and most have an operating wattage and a higher startup wattage. Ideally, a battery bank will be able to meet all of your needs at a time when multiple appliances are running.
Different types of deep cycle lead acid batteries for solar
Here’s where the rubber meets the road. There are three main types of deep cycle lead acid batteries, and each has its own benefits and drawbacks. They include:
Flooded lead acid batteries
Absorbent Glass Mat (AGM) batteries
Gel batteries
The first kind is inexpensive and long-lasting, but requires regular maintenance to keep the electrolyte in balance with the water inside the battery case.
The other two fall under the broader category of “sealed lead acid batteries”. While they are designed to be maintenance-free by sealing the electrolyte inside the case, they're also more expensive and may not last as long.
Flooded lead acid batteries
The least complicated and least expensive kinds of deep-cycle batteries are flooded lead acid (FLA) batteries. These batteries are the most similar to the image of the simple lead acid battery shown below, with cylindrical lead plates submerged in an electrolyte bath of water and acid.
FLA batteries may be inexpensive, but they’re also complicated to care for, because the electrolyte inside the battery case evaporates over time, requiring you to add distilled water at regular intervals. If you don’t add the water often enough, the battery’s lifespan will be greatly decreased.
Because they are built to be opened, FLA batteries need to be stored upright and given adequate ventilation to prevent a buildup of the hydrogen gas that is released when the battery discharges. This means keeping a bank of deep cycle FLA batteries suitable for home energy storage can take up a lot of space, as shown in the image above.
If properly cared for and discharged to no more than half of their capacity on a regular basis, FLA batteries can last from 5 to 8 years in a home energy storage setup.
Sealed lead acid batteries
As the name suggests, sealed lead acid (SLA) batteries cannot be opened and do not require water refills.

A bank of sealed lead acid batteries.
Instead, they use one of two methods to keep nearly all the water present in the electrolyte inside the battery body—either by adding absorbent fiberglass separators between the positive and negative plates or by turning the electrolyte into a gel. These designs mean SLA batteries can be placed on their sides and stacked for easy storage.
Despite these improvements, SLA batteries still need to be vented to prevent hydrogen gas buildup. This vent is controlled by a valve, which is why SLA batteries are sometimes also called “valve-regulated lead acid”, or VRLA batteries.
As referenced above, there are two main kinds of SLA batteries: Absorbent Glass Mat (AGM) and gel batteries.
AGM batteries (Absorbent Glass Mat)
AGM batteries were developed when battery manufacturers were looking for the best way to keep the electrolyte close to the plates for use in high-vibration applications like golf carts.
The mat in this case is made of a fine fiberglass mesh that kind of looks like a gauze pad, and is placed between the negative and positive plates in each of the cells of the battery.
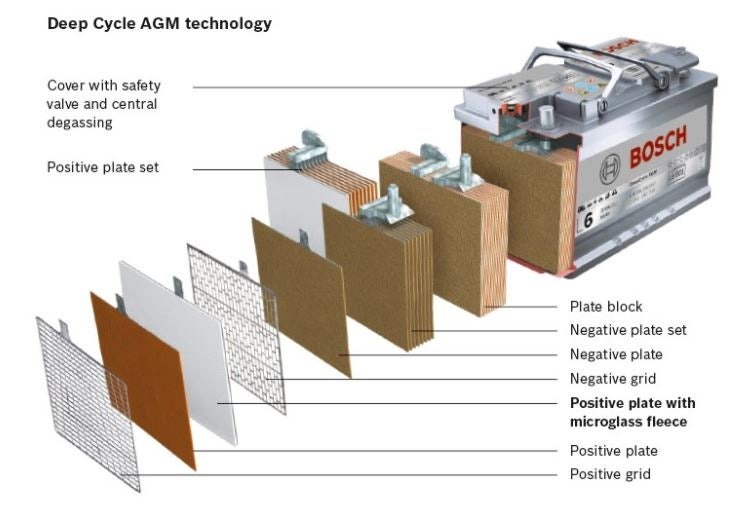
An example showing the interior parts of a typical AGM battery. Image source: Bosch
Because an AGM battery traps nearly all of the electrolyte inside the battery, it can be discharged to a greater depth while allowing the sulfate to recombine with the free hydrogen.
High-quality AGM batteries can be discharged to 80% of their capacity over hundreds of cycles, and typically last 4 to 6 years in a home energy storage setup. Discharging to no more than 50% is still recommended for maximum battery life.
These advantages come at a price, though, and AGM batteries typically cost 1.5 to 2 times as much per kilowatt-hour (kWh) of energy storage. AGM batteries also take up slightly more space per kWh, but again, they can be stacked on their side in order to save space in a home storage setup.
Choose AGM batteries for solar energy storage if you prefer not to maintain a strict schedule of testing and watering FLA batteries, you want versatile mounting options and long life, and you’re willing to pay for it.
Gel batteries
Like AGM batteries, gel batteries are designed to keep the electrolyte inside the battery and prevent it from evaporating or spilling.
In this design, the sulfuric acid is suspended in a silica gel, which makes the battery very stable and has a low self-discharge rate over long periods of time.

An example of a deep cycle gel battery. Image source: Taico
Gel batteries are the safest lead acid batteries because they release very little hydrogen gas from their vent valves. They perform well in places where high temperatures are a concern, and can be discharged below 50% and retain their lifespan.
The drawbacks of gel batteries are that they store less energy in the same space as other lead acid battery types, and the gel doesn’t perform well in low temperatures.
Also, gel batteries require careful charging with specific smart chargers that are designed to limit the voltage used to charge the batteries and prevent overcharging. Failing to use these chargers can result in a battery that dies years before its time.
Choose gel batteries for solar energy storage if you live in a hot climate and can’t store your batteries somewhere cool or well-ventilated, and also if you can absolutely 100% make sure they’re never charged at voltages outside their specific range.
Are lead acid batteries better than lithium ion batteries?
The short answer to this question is no, lead acid batteries are not better than lithium ion batteries.
It is worth noting, however, that lithium ion is a newer battery technology that has specific advantages over lead acid, including:
Greater energy density (more energy in a smaller space)
Higher tolerance for temperature changes
The ability to be regularly discharged to 80% and still last 10 years
For example, a lithium ion battery like the Tesla Powerwall takes up just about 4.5 cubic feet, hangs on a wall, stores 13.5 kWh of usable energy, and has a warranty that says it will last for at least 10 years while still being able to store 70% of its initial capacity. With daily discharge of up to 80%, that’s something like 33,000 kWh served.
Whereas a deep cycle battery bank made up of flooded lead acid batteries that could discharge up to 10.4 kWh per day would take up 8.2 cubic feet on the floor, require regular maintenance, and last for about 7 years total, serving about 28,000 kWh.
Are lead acid batteries cheaper than lithium ion batteries?
There is no such thing as a low-cost battery, but there is a difference in price between these two kinds of batteries, of course.
Buying the lead acid batteries we described above along with all the testing and watering equipment would cost around $7,500 including installation costs. Buying the Powerwall at current prices, would cost $12,850 with installation costs.
Energy storage costs qualify for the federal clean energy tax credit. The tax credit is up to 30% of the cost to install the system. After the tax credit, the lead acid battery system described above would cost $5,250, and the Powerwall costs would be about $8,400.
Dividing the cost by the expected lifetimes, the lead acid costs $750 per year of service, and the Powerwall would cost $900 per year, or 20% more.
Reviews of lead acid batteries
For more information, check our pages on the best solar batteries and Trojan lead acid batteries.
Ben Zientara is a writer, researcher, and solar policy analyst who has written about the residential solar industry, the electric grid, and state utility policy since 2013. His early work included leading the team that produced the annual State Solar Power Rankings Report for the Solar Power Rocks website from 2015 to 2020. The rankings were utilized and referenced by a diverse mix of policymakers, advocacy groups, and media including The Center...
Learn more about Ben Zientara